A new sustainable and practical method for producing hydrogen from water has been discovered by a team of researchers at the RIKEN Center for Sustainable Resource Science (CSRS) in Japan led by Ryuhei Nakamura. Unlike current methods, the new method does not require rare metals that are expensive or in short supply. Instead, hydrogen for fuel cells and agricultural fertilizers can now be produced using cobalt and manganese, two fairly common metals. The study was published in Nature Catalysis.
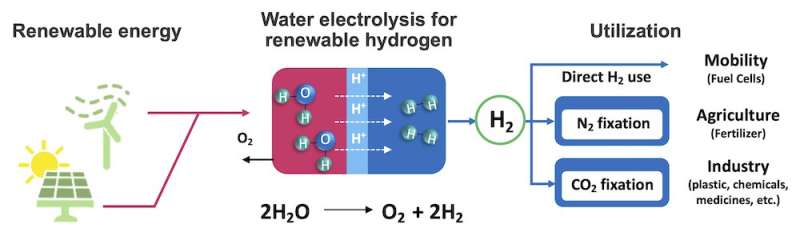
Unlike conventional fossil fuels that generate carbon dioxide upon combustion, hydrogen is a clean fuel that only produces water as a byproduct. If hydrogen can be extracted from water using renewable electricity, the energy grid can be made clean, renewable, and sustainable. Additionally, hydrogen is the key ingredient needed to produce ammonia, which is used in virtually all synthetic fertilizers. But instead of cleanly extracting hydrogen from water, currently, ammonia plants use fossil fuels to produce the hydrogen they need.
- So why are we still using fossil fuels? One reason is that the hydrogen extraction process itself — electrolysis — is expensive and not yet sustainable.
“This is primarily due to a lack of good catalysts. Currently, the most active catalysts for water electrolysis are rare metals like platinum and iridium, which creates a dilemma because they are expensive and considered “endangered species” among metals.
Switching the whole planet to hydrogen fuel right now would require about 800 years’ worth of iridium production, an amount which might not even exist. On the other hand, abundant metals such as iron and nickel are not active enough and tend to dissolve immediately in the harsh acidic electrolysis environment.
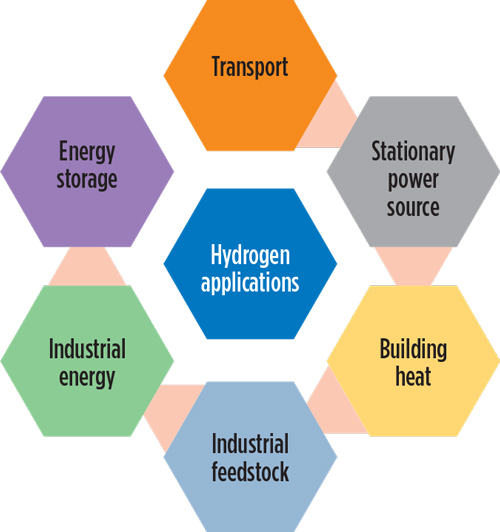
Why do we need hydrogen?
Energy efficiency, renewable power, and direct electrification can reduce emissions from electricity production and a portion of transportation; but the last 15 percent or so of the economy, comprising aviation, shipping, long-distance trucking and concrete and steel manufacturing, is difficult to decarbonize because these sectors require high energy density fuel or intense heat. Green hydrogen could meet these needs.
In their search for a better catalyst, the researchers looked at mixed cobalt and manganese oxides. By combining them, the researchers hoped to take advantage of their complementary properties. It was necessary to consider the high current density needed for practical application outside the laboratory.
For industrial scale hydrogen production, it was needed to set study’s target current density to about 10 to 100 times higher than what has been used in past experiments,” “The high currents led to a number of problems such as physical decomposition of the catalyst.”
Eventually, the team overcame these issues by trial and error, and discovered an active and stable catalyst by inserting manganese into the spinel lattice of Co3O4, producing the mixed cobalt manganese oxide Co2MnO4.
Testing showed that Co2MnO4 performed very well. Activation levels were close to those for state-of-the art iridium oxides. Additionally, the new catalyst lasted over two months at a current density of 200 milliamperes per square centimeter, which could make it effective for practical use. Compared with other non-rare metal catalysts, which typically last only days or weeks at much lower current densities, the new electrocatalyst could be a game changer.
In the long run, scientists are believing that this is a huge step towards creating a sustainable hydrogen economy. Like other renewable technologies such as solar cells and wind power, it is expected that the cost of green hydrogen technology to plummet in the near future as more advances are made.”
The next step in lab will be to find ways to extend the lifetime of the new catalyst and increase its activity levels even more.
You must be wondering why there is so much content and news on Hydrogen? We can understand your feelings! Now if you have already spent so much time on this blog then please hold for few more minutes and you would get to know about whole scenario and why this single chemical element is getting so much importance.